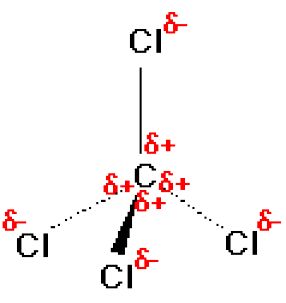
Which of the following is a polar molecule? (1) Para dichlorobenzene (2) Carbon tetrachloride (3) Tetrachloroethene (4) HO benzeneOH
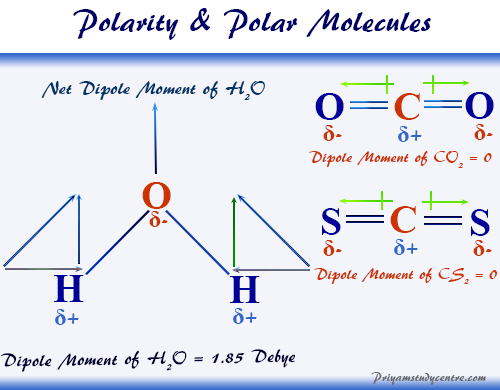
Polarity of Molecules 11/18/14 Polar Molecules are molecules which have an uneven distribution of charge. One side of the molecule is negative while. - ppt download
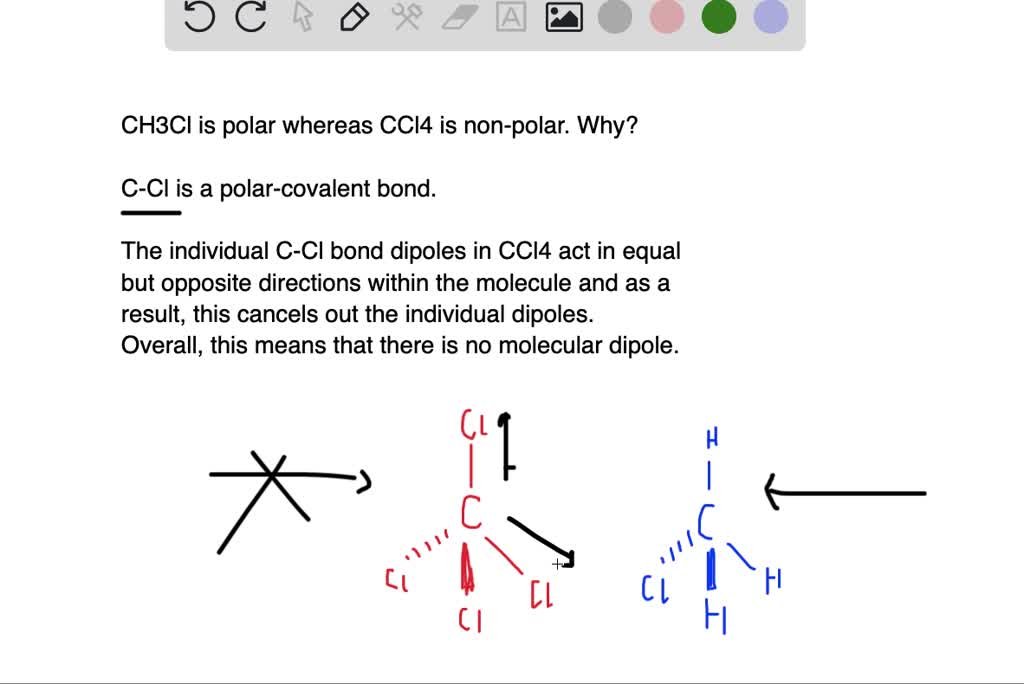
SOLVED:Account for the fact that chloromethane, CH3 Cl, which has only one polar C-Cl bond, is a polar molecule, but carbon tetrachloride, CCl4, which has four polar C-Cl bonds, is a nonpolar

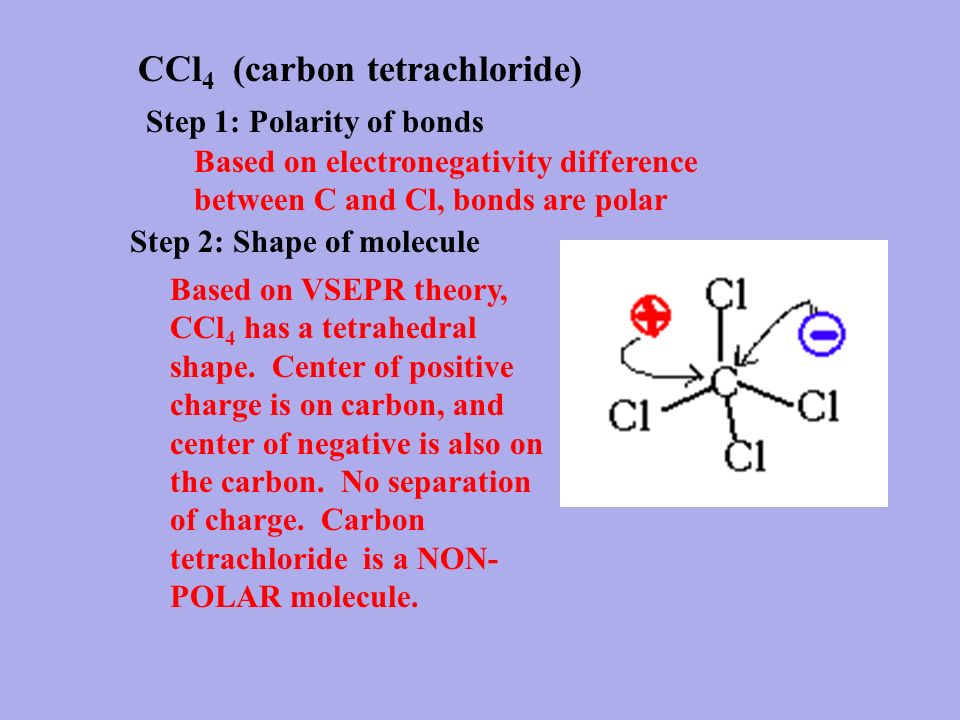
Carbon Tetrachloride (CCl4); Lewis Structure, Molecular Geometry, Polarity, And Applications | Scientific Sarkar

Which solvent, water or carbon tetrachloride, would you choose to dissolve each of the following? a. KrF_2 b. SF_2 c. SO_2 d. CO_2 e. MgF_2 f. CH_2O g. CH_2=CH_2 | Homework.Study.com
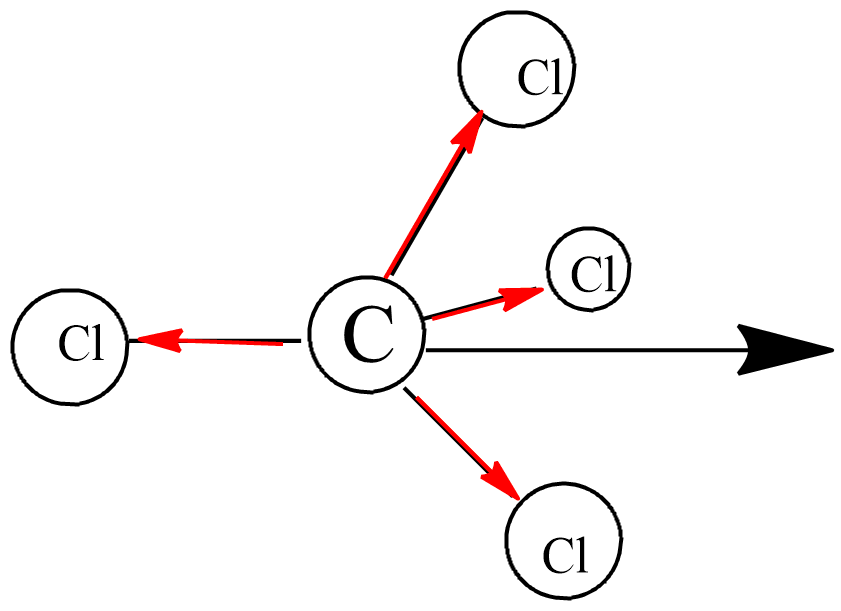
Carbon tetrachloride ($CC{{l}_{4}}$ ):(A) ionic substance(B) non-polar covalent substance(C) polar covalent substance (D) macromolecular substance(E) metallic substance
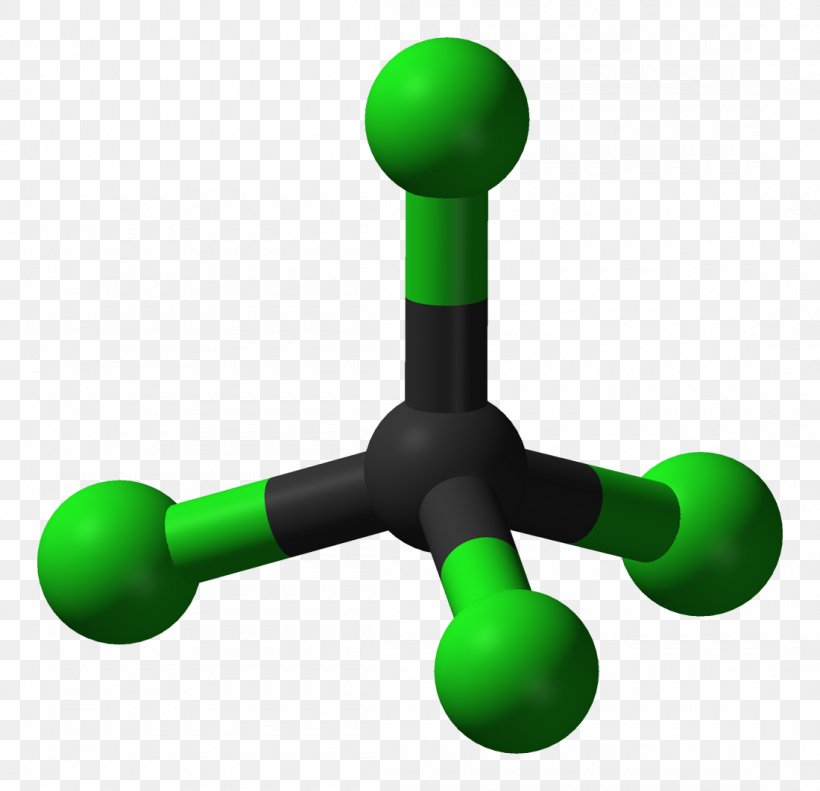
Carbon Tetrachloride Molecule Carbon Dioxide Chemical Polarity, PNG, 1100x1062px, Carbon Tetrachloride, Carbon, Carbon Dioxide, Carbon Disulfide,

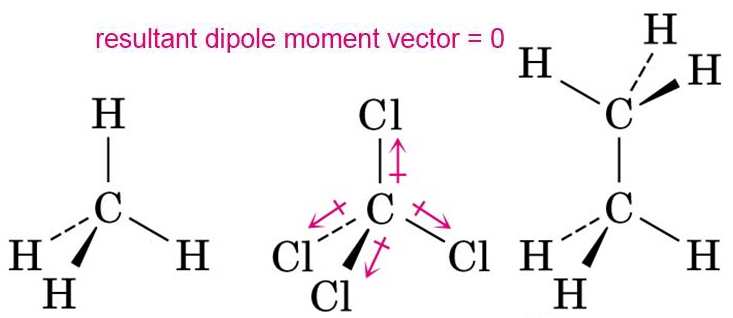





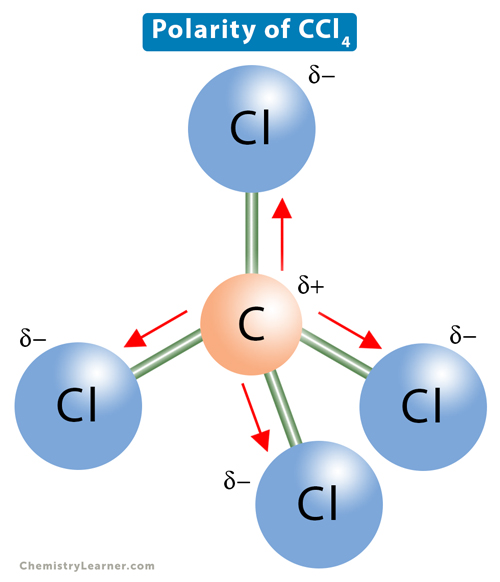






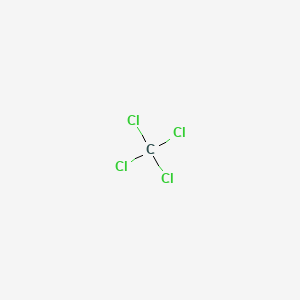


![Is \\[CC{l_4}\\] polar or nonpolar? Is \\[CC{l_4}\\] polar or nonpolar?](https://www.vedantu.com/question-sets/dbf3f5ee-35e7-43b7-b8a3-cfe63dfd520e2097468885289678636.png)