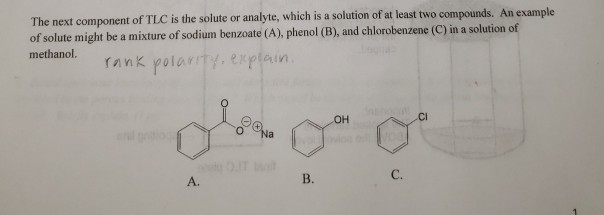
Explain why the dipole moment of chlorobenzene is lower than cyclohexyl chloride ? – The Unconditional Guru

Table 1 from A dispersive liquid-liquid microextraction using a switchable polarity dispersive solvent. Automated HPLC-FLD determination of ofloxacin in chicken meat. | Semantic Scholar

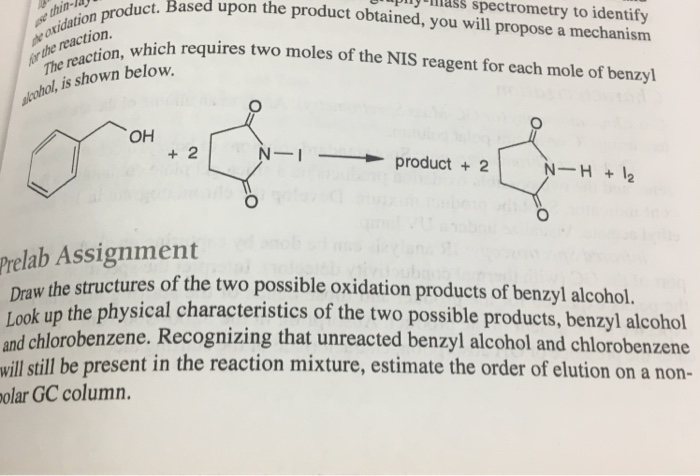
The elution sequence of a mixture of compounds containing chlorobenzene, anthracene and p-cresol developed on an alumina column using a solvent system of progressively increasing polarity is (a) anthracene to chlorobenzene to
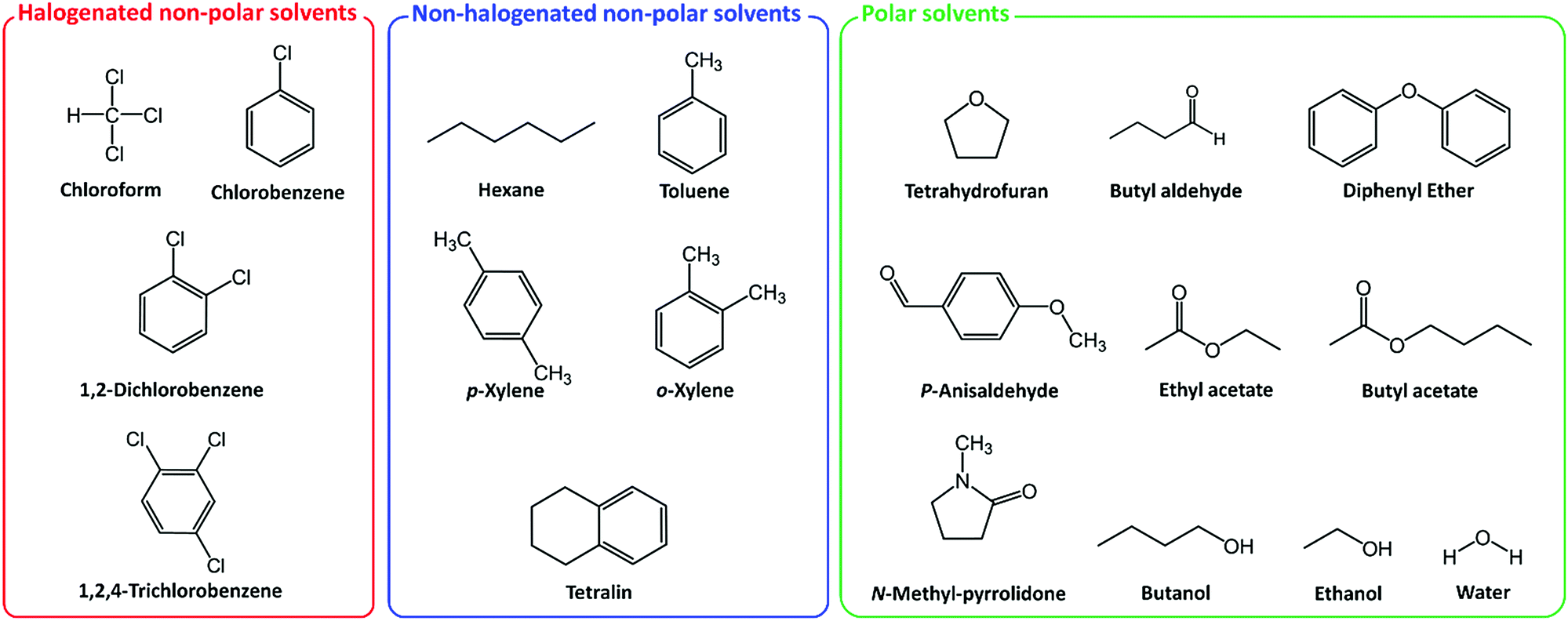
Environmentally benign fabrication processes for high-performance polymeric semiconductors - Journal of Materials Chemistry C (RSC Publishing) DOI:10.1039/C6TC05535D

Explain why (i) The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride?(ii) Alkyl halides, though polar, are immiscible with water?(iii) Grignard reagents should be prepared under anhydrous conditions?